This order makes qualitative sense based on the kinds and extents of motion available to atoms and molecules in the three phases. K), whereas S° for water vapor is 188.8 J/(mol.For instance, S° for liquid water is 70.0 J/(mol Excited state at 3.0 eV,81WU /TIE.\), for substances with approximately the same molar mass and number of atoms, S° values fall in the order S°(gas) > S°(liquid) > S°(solid). 
Gas phase Relative to HBr, reevaluated with current HBr acidity. Gas phase Entropy change calculated or estimated Mīy formula: NO 3 - + H + = HNO 3 Quantity Reaction search pages in place of the enumerated reaction Go To: Top, Gas phase thermochemistry data, Phase change data, Henry's Law data, IR Spectrum, Vibrational and/or electronic energy levels, References, Notes Chickos Enthalpy of vaporization Δ vapH (kJ/mol) 
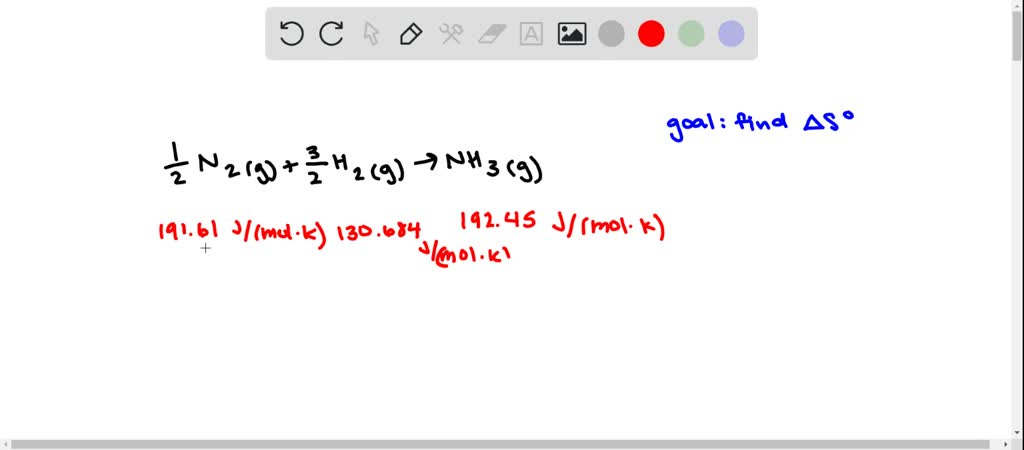
Go To: Top, Gas phase thermochemistry data, Reaction thermochemistry data, Henry's Law data, IR Spectrum, Vibrational and/or electronic energy levels, References, Notesĭata compiled by: William E. Requires a JavaScript / HTML 5 canvas capable browser. Gas Phase Heat Capacity (Shomate Equation) Secretary of Commerce on behalf of the U.S.A. In case of this change in a reaction the symbol will become H. Go To: Top, Phase change data, Reaction thermochemistry data, Henry's Law data, IR Spectrum, Vibrational and/or electronic energy levels, References, Notesīy the U.S. The symbol of standard enthalpy change is Delta H nought or H. For example, the entropy change for the vaporization of water can be found as follows: So So(H 2O(g)) So(H 2O(l)) 188.7J/K mol 69.9J/K mol 118.8J/K mol. Your institution may already be a subscriber.įollow the links above to find out more about the data In chemistry, the standard molar entropy is the entropy content of one mole of substance, under standard conditions (NOT STP). The knowledge of the absolute entropies of substances allows us to calculate the entropy change (So) for a reaction. With the development of data collections included in The purpose of the fee is to recover costs associated NIST subscription sites provide data under theĭata Program, but require an annual fee to access. For N2H4(l) in the interval from 1.53☌ to 298.15K, the molar heat capacity at constant pressure is given by the expression Cp 97.8 + 0.0586(T 280).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
